Table of Contents Show
Crystals are a common everyday occurrence. In modern human society, we interact with some form of crystals at almost all times of the day. Yet it may not entirely be clear what crystals really are.
But if you feel that crystals are just shiny gemstones used to manufacture stunning jewelry, you couldn’t be more wrong. In fact, that is one of the least common uses of crystals.
What are crystals?
Crystals are scientifically defined as solids that have a regular three dimensional arrangement of particles in them. Every solid object in the universe is made of tiny particles. An object in which these tiny particles are placed in a regular, repeating arrangement, is referred to as a crystal.
These tiny particles are held together by strong forces of attraction which keep the crystal structure stable. The difference in this attractive force amongst different crystals is the basis of the different types of crystals that exist.
There are four different types of crystal structure we know so far. They are separated by the arrangement of molecules and bonds within them. You can also skip to the chemistry here.
8 Types of Crystals and their benefits
It is not uncommon, even in today’s modern world, to see certain crystals being sold for their incredible, life-altering benefits. Historically, it is unclear when exactly these beliefs started to manifest but Greek artifacts often display certain crystals used for various spiritual purposes.
Following are eight of the most common crystals and their connections to spiritual chakras as well as their benefits:
1. Amethyst

For good reason, this is one of the most popular crystals. The purple-hued crystal’s effects are said to vary from calming anxiety to ensuring a good night’s sleep.
Amethyst acts to create physical and emotional peace by focusing on the crown chakra (placed at the very top of your head and associated with the mind).
Its specialized medicinal characteristics include headache and migraine treatment, pain reduction, and intuition strengthening.
Many people think that keeping an amethyst geode near your bed may help you sleep better.
2. Rose Quartz

This pinkish stone is excellent for creating love and new connections. If you’re already on an emotional rollercoaster, rose quartz promotes reconciliation and understanding.
Another advantage? It is claimed to provide inner serenity regardless of your relationship status.
3. Black Tourmaline

The absorbent crystal is supposed to be capable of absorbing all bad vibrations that come your way, therefore eliminating toxicity.
Black tourmaline, which is associated with the root chakra, is said to anchor you when life gets overwhelming.
4. Selenite

Are you constantly exhausted? It may be time for an aura cleanse (in addition to another cup of coffee and some serious self-care). Selenite is the ideal gemstone for revitalizing our auric field.
The crystal is said to wipe away the day’s negative vibrations and immerse you in a more calm flow of energy. Use selenite as a wand over your body to assist pull away all the energy impediments to achieving inner tranquility.
Keep in mind that selenite dissolves in water, so make sure the geode stays dry.
5. Citrine

Citrine, a brilliant yellow quartz type, is thought to ease individuals who are stuck at a difficult point in their lives.
While relaxing, place citrine on your belly or solar plexus chakra for a quick boost of confidence. The stone is said to untangle the tangled energy within and restore pleasant vibrations.
6. Jade

We could all use a little luck in our lives, which is why jade is so popular: It functions as a portable fortune charm.
Jade is thought to bring wealth and success in both the financial and social realms. Wearing jade on your left hand or wrist is supposed to assist in cosmic favors. Jade rings, on the other hand, are pretty attractive too!
7. Clear Quartz

Clear quartz, a versatile necessity for every crystal collection, will help you “reboot” your life.
Clear quartz, sometimes known as a “master” crystal, is all about reducing inner noise and focusing your goals. The well-known geode is said to have all-purpose therapeutic powers.
Place your new clear quartz near a window to absorb sun energy, which will then be transferred to you and maybe light away negative energy.
8. Rhodochrosite

Those who believe in crystal healing, feel that rhodochrosite is the stone that was virtually built for breakup rehabilitation.
While meditating, use rhodochrosite, which stimulates the heart chakra, to increase self-love and confidence. The pink crystal may perhaps set you up for the next love experience by instilling an optimistic mindset.
Spiritual Properties of Crystals
While the scientific information on crystals has recently found massive ground as part of a branch of physics called solid state physics, they have historically been used by humans for much longer.
Ancient scripts and portraits often mention crystals and their uses as spiritual artifacts. These spiritual artifacts have been historically used for meditation and spiritual blessings.
It is not uncommon even in today’s modern world to see certain crystals being sold online or otherwise for their incredible life altering benefits.
Historically, it is unclear when exactly these beliefs started to manifest but Greek artifacts often display certain crystals used for various spiritual purposes.
It was perhaps always a human desire to have a physical connection with a spiritual object that led to the rise of popularity of using healing crystals.
These crystals are now known to be used for almost all ailments. It is said that they absorb negative energy and some even emit positive energy which lifts peoples spirits and makes them happy.
Believers of this branch of science claim that these crystals are associated with certain chakras in our bodies and souls. Chakras are spiritual points in our bodies that influence the flow of energy inside us.
The stones associated with each chakra has the ability to allow energy to flow through it with ease. This in turn relieves stress and cures many ailments.
If you would like to know the science behind crystals, read on!
Different Chemical Structures Found in Crystals
Various different objects may be made up of different particles. These atomic particles as they are referred to are often one or more of the following; atoms, ions, or molecules.
The presence and arrangement of the particular atomic particles in an object determines the type and properties of the attractive force present in a crystal.
First and foremost, all crystals are differentiated based on their crystalline structure which is influenced by the attractive force that results in the particular crystalline structure. The different crystal structures are:
1. Ionic lattice structures
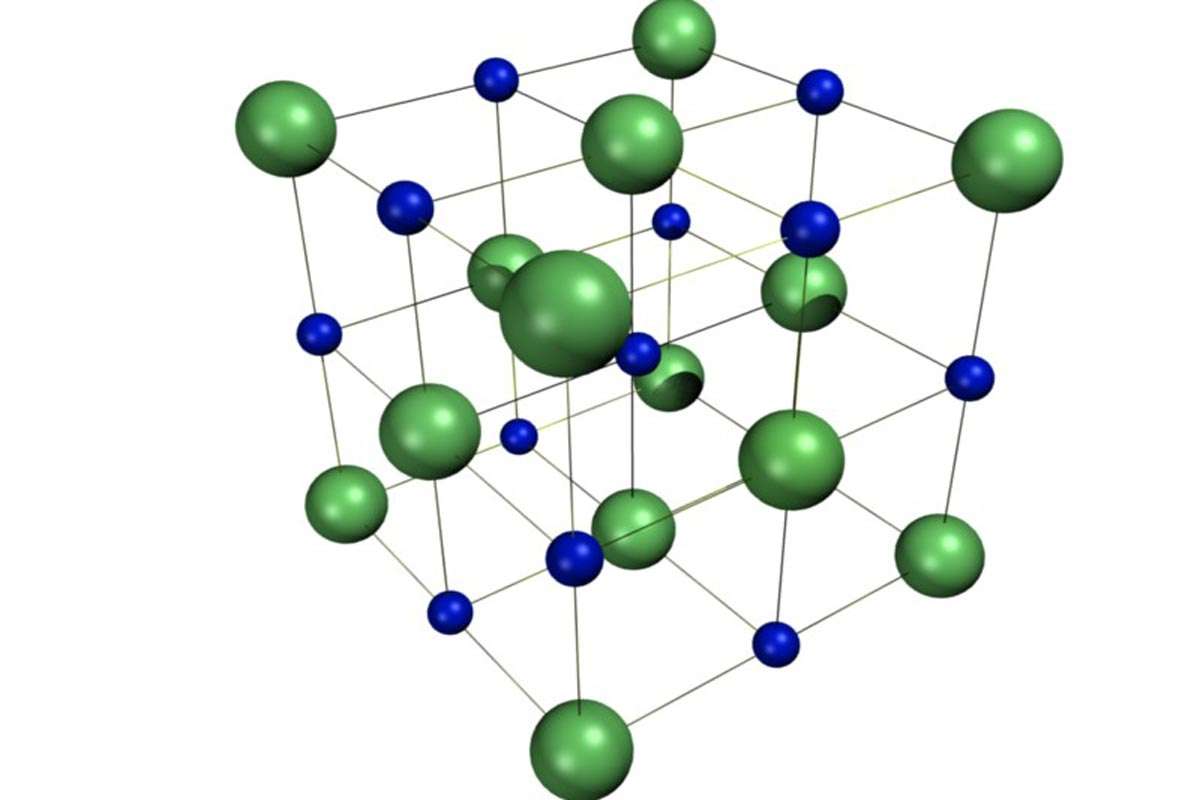
Ionic crystals have a giant ionic lattice structure. This is basically a three dimensional regular structure made of alternating charged particles. These charged particles are called ions.
Ions are tiny particles that have either a positive electric charge or a negative electric charge. These positive and negative charges have a fundamental property of attracting each other.
So while a positive charge will attract a negative charge, it will also repel another positive charge. This attraction between two opposite charges is known as electrostatic attraction and the force responsible for this is called the electrostatic force of attraction.
It is this force that creates the ionic crystalline lattice structure. This ionic force, as it is sometimes called, is very strong and the bond formed between particles due to this force is as a consequence extremely strong.
In an ionic lattice structure there is an alternating, regular arrangement of positive and negative charges in all three dimensions. This repeating arrangement can often be billions of particles long which is why this structure is also called the giant ionic lattice structure.
A common example of an everyday substance with this structure is table salt. A fun fact about table salt is that each salt grain is made up of millions of ions. That’s how small ions really are.
2. Molecular Crystalline Structures
Molecular crystals are somewhat similar to ionic solids. They have a similar regular crystalline structure. However, in molecular crystals the fundamental particles involved are not ions but molecules instead.
These molecules can be of various types. The type of molecule present in bonding is what determines which force binds the molecular crystal.
In both cases of molecular solids, the molecules form bonds with each other to form a regular three dimensional arrangement almost exactly the same way as ionic lattices do.
The only difference is the strength of the bonds in the two types of crystals. An example of common molecular crystalline structures is dry ice which is also known as solid carbon dioxide.
Usually the molecules are either polar or nonpolar. The polarity of a molecule is described as a small or fractional charge on a part of the molecule.
In simple terms this means that a polar molecule is one which has a slight positive charge on one side and a slight negative charge on the opposite side. Such molecules are called dipoles and form bonds which are referred to as dipole-dipole bonds.
3. Network Covalent Crystalline Structure
Network Covalent Crystals are also called giant covalent crystals. These crystalline structures are in fact made up of atoms bonded together.
However, unlike in the previous examples the atoms in this structure aren’t bonded by ionic bonds. There are no positive and negative charges to begin with so there is no electrostatic force to bind the atoms.
Giant covalent structures are instead bonded by a different bond called the covalent bond. Here, instead of being attracted to each other, the microscopic atoms are attracted to common points between them.
Each covalent bond is therefore likewise an attraction of two atoms to the approximately central point between them. As a result these bonds individually are not exactly very strong.
However, when a network of such bonds is formed within an object, it can drastically increase the strength of the material. An excellent example of this is the arrangement of carbon in diamond.
Diamond is a network covalent crystal that is entirely made up of carbon atoms. In diamond each carbon atom is attached to four other carbon atoms, which are then attached to a further four carbon atoms.
In this manner an expanding network is formed. The sheer number of bonds present in these giant structures is what is responsible for their incredible strength.
4. Metallic Crystalline Structures
Metallic Crystalline structures are a bit unique. These crystalline structures are linked together like large metallic sheets.
In these crystals there are positively charged ions surrounded by what is described as a sea of negatively charged electrons. Electrons are very small negatively charged particles. Electrons are more than a thousand times smaller than atoms or ions.
The positively charged ions are attracted to the negatively charged electrons. Due to the large number of electrons present there are plenty of bonds formed in this structure as well which gives metallic crystals their strength.
Metallic crystals are strong but also malleable. This means that they can be easily bent and made into any shape desired. Most metals are examples of this crystalline structure. Copper is probably the most common example of a metallic crystal in everyday life.






